- Bone Health
- Immunology
- Hematology
- Respiratory
- Dermatology
- Diabetes
- Gastroenterology
- Neurology
- Oncology
- Ophthalmology
- Rare Disease
- Rheumatology
FDA Approves Celltrion’s Yuflyma, the Ninth Adalimumab Biosimilar
Celltrion Healthcare announced that the FDA has approved Yuflyma (adalimumab-aaty) as the ninth biosimilar referencing Humira (adalimumab).
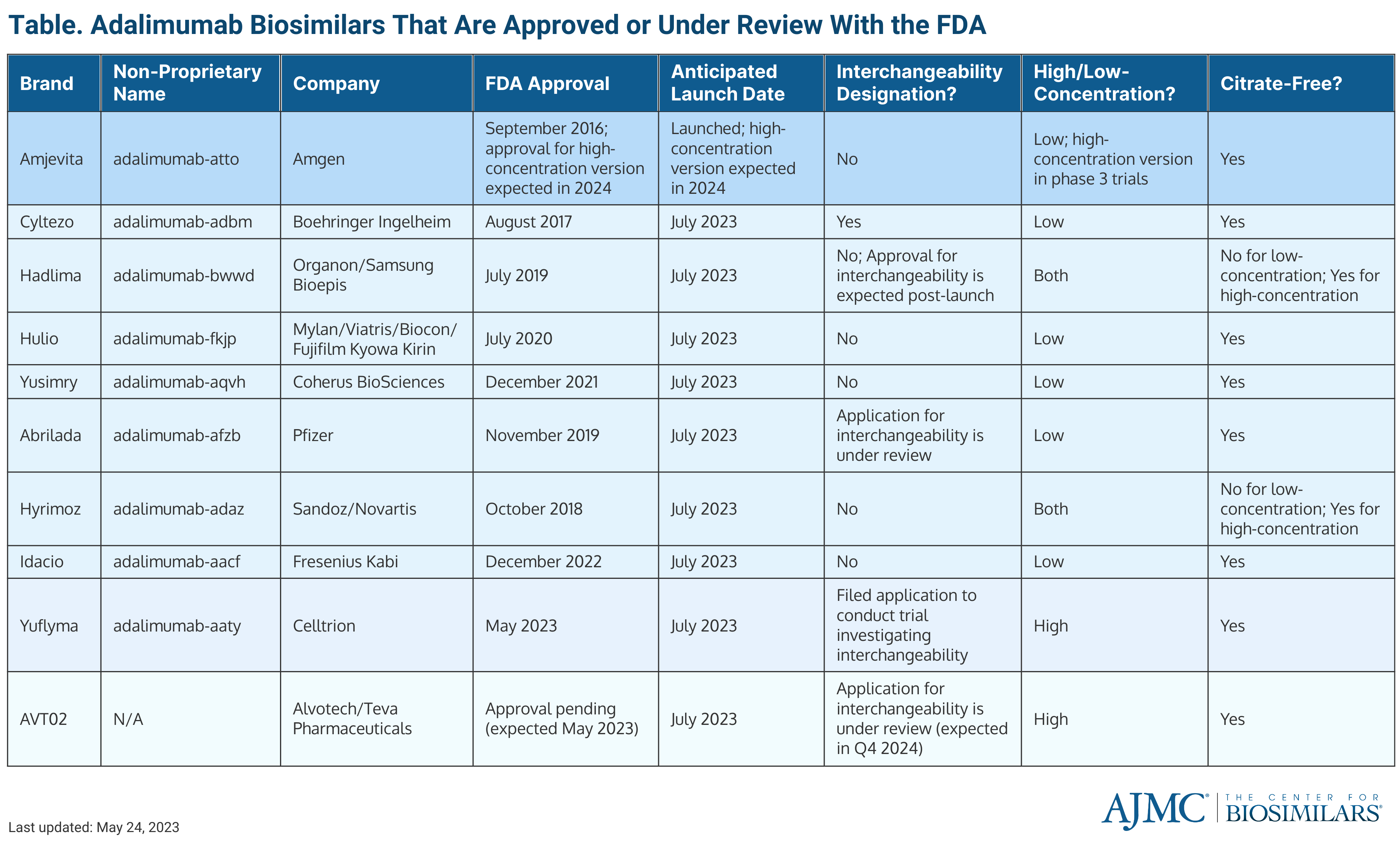
Celltrion Healthcare announced the FDA approval of its adalimumab biosimilar, Yuflyma (adalimumab-aaty), making it the ninth biosimilar referencing Humira (adalimumab) to receive regulatory approval in the United States (Table).
Yuflyma is a high-concentration (100mg/mL), citrate-free adalimumab biosimilar. The biosimilar will be offered to patients in a prefilled syringe and autoinjector administration options.
Click to enlarge
“Currently, more than 80% of patients treated with Humira in the United States rely on a high-concentration and citrate-free formulation of this medication. The availability of a high-concentration and citrate-free formulation adalimumab biosimilar provides an important treatment option for patients with inflammatory diseases who benefit from this effective therapy,” said Jonathan Kay, MD, a professor of medicine and the associate director of the MD/PhD Program at the University of Massachusetts School of Medicine, in a statement from Celltrion.
The FDA approved the biosimilar for 8 indications:
- rheumatoid arthritis
- juvenile idiopathic arthritis
- psoriatic arthritis
- ankylosing spondylitis
- Crohn disease
- ulcerative colitis
- plaque psoriasis
- hidradenitis suppurativa
Yuflyma is the fifth biosimilar and second anti-tumor necrosis factor biosimilar developed by Celltrion Healthcare to receive FDA approval. Yuflyma is also approved in the European Union and Canada.
“Yuflyma offers patients a high-concentration and citrate-free formulation of adalimumab biosimilar, providing an alternative treatment option for patients. It represents a key treatment option in patient care and patient choice…. As a leading global biopharmaceutical company, we are leveraging our unique heritage in biotechnology, supply chain excellence, and best-in-class sales capabilities to expand the availability of high-quality biosimilars for U.S. patients,” commented Tom Nusbickel, chief commercial officer at Celltrion USA, in a statement.
Celltrion said that Yuflyma will launch on the US market in July 2023. The company said that it is also seeking an interchangeability designation for the biosimilar, predicting that the extended approval will come in the fourth quarter of 2024.
The news comes after the US welcomed the first adalimumab biosimilar on the market, Amgen’s Amjevita, in January 2023. It will be the only adalimumab biosimilar competing against the originator until July 2023.
Additionally, Boehringer Ingelheim announced that the FDA approved the Cyltezo Pen, a new autoinjector option for the company’s adalimumab biosimilar (Cyltezo; adalimumab-adbm) on May 22, 2023. Cyltezo was initially approved as a pre-filled syringe. The 40 mg/0.8 mL pre-filled Cyltezo Pen will be available in 2-, 4- and 6-pack options on July 1, 2023. Cyltezo is also the only adalimumab biosimilar to have an interchangeability designation so far.
In March 2023, the FDA approved the high-concentration, citrate-free formulation of Sandoz’ Hyrimoz (adalimumab-adaz).
Across 2023, an estimated 10 adalimumab biosimilars will enter the US market, with 9 expected to launch in July 2023. In addition to the aforementioned adalimumab biosimilars, the other FDA-approved adalimumab products are Idacio, Abrilada, Yusimry, Hadlima, and Hulio.
The FDA is also reviewing Alvotech’s adalimumab biosimilar, AVT02, which recently received a second complete response letter (CRL) that cited “deficiencies” in the company’s Iceland-based manufacturing facility. The CRL pushed back the FDA’s decision date for approval from April 2023 to later June 2023.
Newsletter
Where clinical, regulatory, and economic perspectives converge—sign up for Center for Biosimilars® emails to get expert insights on emerging treatment paradigms, biosimilar policy, and real-world outcomes that shape patient care.
